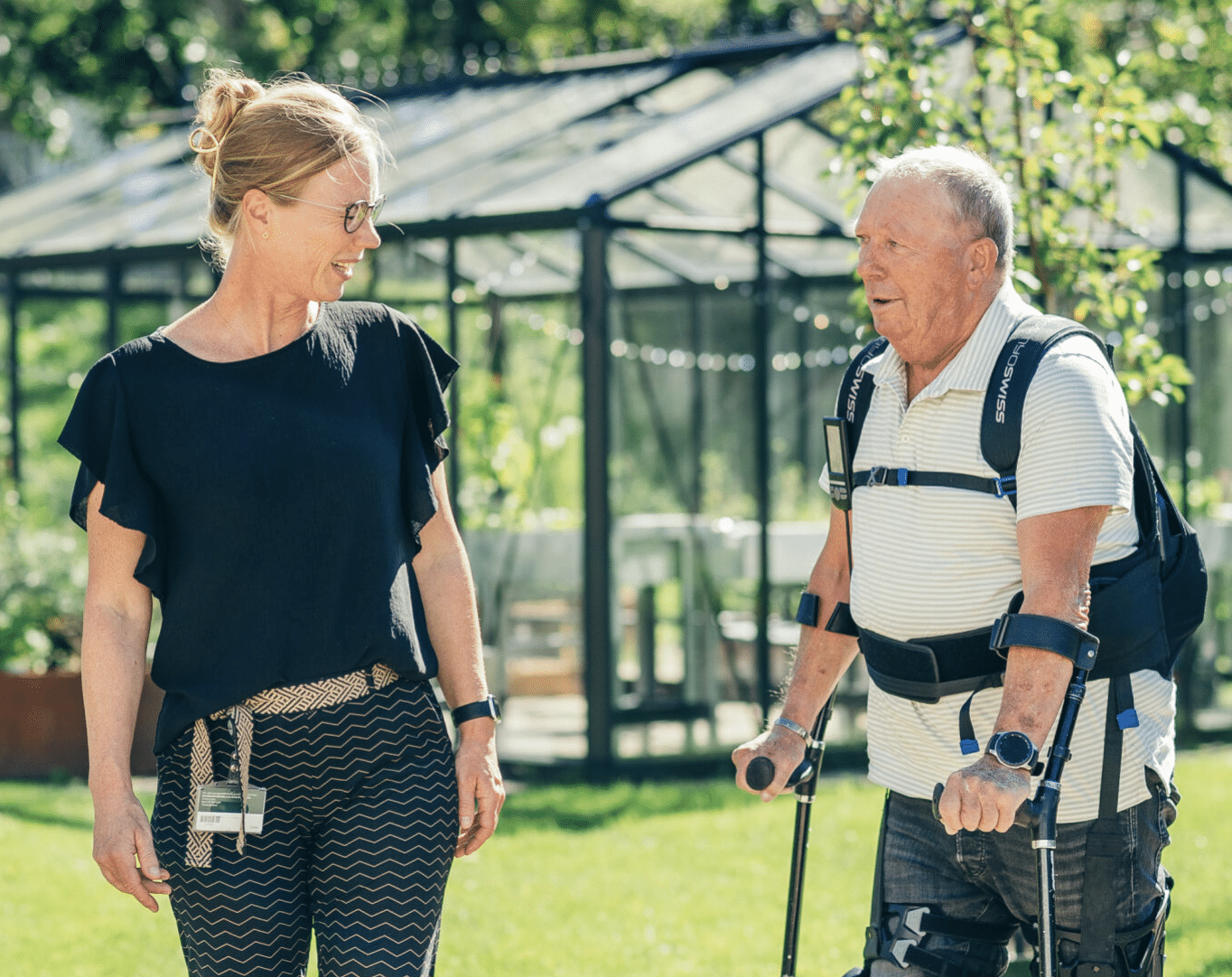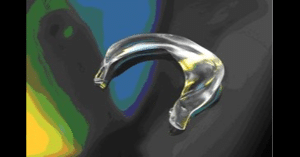- Featured story
Newsroom
Standing on the shoulders of giants! Our editorial office is dedicated to sharing the stories, knowledge and experiences of innovators in Health & High Tech. Discover their journey, struggles and victories as you swipe through the podcasts, clips and articles in our newsroom. Enjoy!
News
- 2025-06-18
Saskia Eijkelhof – Our New Community Development Manager as of July 1, 2025
We are pleased to announce that Saskia Eijkelhof will join Noviotech Campus as Community Development Manager starting July 1, 2025....- 2025-03-26
Hans Schoonen nieuwe Algemeen Directeur Noviotech Campus per 1 april 2025
Per 1 april 2025 is Hans Schoonen benoemd tot Algemeen Directeur van Noviotech Campus. Met zijn uitgebreide ervaring op het...- 2025-01-22
A hemostatic product for the operating room
A hemostatic product for the operating room An early Christmas thought in 2009 makes Johan Bender found the company GATT....- 2025-01-22
The RobotLab Program by Big Chemistry establishes at Noviotech Campus!
The RobotLab Program by Big Chemistry establishes at Noviotech Campus! Big Chemistry: A big data approach to chemical reactions The...- 2024-12-12
Noviotech Campus uitgeroepen tot Ambassadeursterrein van Werklandschappen van de Toekomst
Noviotech Campus klaargestoomd tot Werklandschappen van de Toekomst Noviotech Campus in Nijmegen behoort samen met twee andere Gelderse bedrijventerreinen tot...- 2024-12-04
Noviotech Campus: Top 13 Innovation Hub in the Netherlands!
Noviotech Campus has officially joined the National Campus Network, securing its position as one of the 13 leading mature campuses...Get funded by Engaged Business Angels from Oost NL
Get funded by Engaged Business Angels from Oost NL Looking for funding from an investor who’s hands-on and fully engaged?...Discover, Connect and Innovate at The Vibe of the Future Festival
Article published by INTO Nijmegen | Lunya van Wunnik The Vibe of the Future Festival is about to transform Nijmegen...Roadworks next to Noviotech Campus
Roadworks are scheduled on Neerbosscheweg, next to Noviotech Campus, from August to November 2024. This may cause some disruptions in...June 20 opens an Exclusive Pop-Up Venue at Noviotech Campus!
Unleash the Ultimate Summer Experience: Exclusive Pop-Up Bar & Food at Noviotech Campus! Book now! On June 20 Strandbar Stek...Economic mission to the semiconductor industry and photonics to the US and Canada
Are you internationally active in the semiconductor industry or in photonics? And are you curious about your business opportunities in…
Entrepreneus delegation trip: Artificial Intelligence in Baden-Württemberg (11-12 June)
The state of Baden-Württemberg is a high-tech region with a high level of innovation. In this region, companies and knowledge…
Foodtruck Arocho’s American Kitchen available on Thursdays at Noviotech Campus
Starting from May 16th, Collin will be ready to serve you a delicious lunch from 11:30 AM to 1:30 PM…
Rabolounge at ASM Student Festival
ASM Student Festival Prior to the ASM festival, the ASM Student Festival will take place on May 17th in Arnhem….
Masterclass: AI for Business
4th of June 2024, 18.00 Masterclass: AI for Business Artificial intelligence (AI) allows you to make machines do things that…
AI enables a more humane healthcare system
Jaap Kroes: "AI enables a more humane healthcare system." Nijmegen tops the international rankings of cities using artificial intelligence to...A students’ perspective on AI: Spence van Asperdt
“There are people who are afraid of AI. But in my opinion, it is a very usable tool, which forces...AI is making us lazy
Pim Haselager: “AI is making us lazy.” According to Pim Haselager, Radboud University Professor of Societal Implications of Artificial Intelligence...Inspiration festival for innovators in Health & High Tech
- The Vibe of the Future Festival - Experience the world of tomorrow at The Vibe of the Future...Subsidy Opportunities for Innovative SME Entrepreneurs in East Netherlands
40 million euros available Do you want to innovate sustainably, test your innovation, or demonstrate it? Now is a good...Can we fight malaria with AI?
Completing an escape room in Nijmegen with top Google researchers. Who can say they did that? Koen Dechering got AI…
We raised € 3.350,- for 3FM Serious Request and ALS!
We are very proud of all of you in raising the amount of a whopping € 3.350,-. Great that we...Moore will never die
In 1965, Intel co-founder Gordon Moore observed that the capacity of computer chips doubles every two years; not only describing…
Valerie van Zuijlen: “You have to go all in.”
What is the key to success? Is it about talent, intelligence or creativity – and what about your resources, like…
Shooting for the moon: the motivation and mindset of Marc Klein Wolt
“Once I set myself a goal, I’m very dedicated and will do everything in my abilities to achieve it.” Radboud…
Two sides to the story: Part 1 – Back on my feet
“When Ton Arts comes out of anaesthesia after surgery, he can only move his head. The rest of his body…
Two sides to the story: Part 2 – Robotic Rehab
“It is a fantastic time to be a rehabilitation physician”, says Ilse van Nes. “We can now use technology to…
Changes in Management and Board of Noviotech Campus
The board of Stichting Ontwikkeling Noviotech Campus and Bert Krikke, director of Noviotech Campus BV, have mutually agreed to part...Nexperia joins United Nations Global Compact Initiative
Noviotech Campus-based Nexperia, the expert in essential semiconductors, is pleased to announce that it has joined the United Nations Global…
Recap of celebrating Radboud 100 years
Between 8 and 14 May, our partner Radboud University organised several activities to celebrate their 100th anniversary this year. Check…
Enzyre: towards market leadership in the blood coagulation
Noviotech Campus-based Enzyre, the spin-out from Radboudumc, has a mission: develop ambulatory diagnostic technology for blood clotting tests that allow…
Recap meet-up SEMICON and Innovation Mission Taiwan
On June 7, High Tech NL held a meet-up for those interested in joining SEMICON Taiwan from 6-8 September and/or…
Partner in the picture: Briskr
‘Stimulating innovative entrepreneurship’. This goal led to a close partnership between Oost NL and Briskr almost six years ago. Whereas…
‘Chinese’ Nexperia won’t get European grant
Chipmaker Nexperia, which has its headquarters at Noviotech Campus in Nijmegen, is not eligible for a grant programme meant to…
10% less contrast medium per patient
Administering contrast fluid to patients is necessary to detect tumours via CT scans, for example. However, these fluids end up…
Radboud University performs better in SDG ranking
The THE Impact Ranking is a ranking that assesses universities based on their contribution to the Sustainable Development Goals (also…
CITC’s infrastructure expanded with confocal scanning acoustic microscope
CITC’s lab has been expanded with a confocal scanning acoustic microscope (CSAM) from acoustic microscopy expert PVA TePla. The microscope…
NXP’s only chip factory in the Netherlands: old, but very efficient
Europe is pumping billions into strengthening its own chip industry. Much attention goes to the smallest, most powerful semiconductors, but…
Queen Máxima visited NXP for the celebration of 70 years of chip production
On 25 May Her Majesty Queen Máxima visited NXP Semiconductors at Noviotech Campus in Nijmegen. The company won the Koning Willem I...Coming up: Meet & Eat ”International” – 6 June 2023
Do you have any idea know how many different nationalities we have at Noviotech Campus? A lot! Come and meet…
MKB Datalab-Oost is official!
Now it is official the first MKB Datalab-Oost! This new lab supports SME entrepreneurs in the east of the Netherlands…
Podcast Bert Krikke
Bert Krikke shines in the Nijmegen Entrepreneurs Podcast . Produced by Raoul Toet & Mark Schaap of Zima Blue. Listen…
Full program available for Benelux RF Conference
Full program available Check out the program of the Benelux RF Conference 2023, which Noviotech Campus is a partner of....Kick-off RF Knowledge Lab at RF Benelux Conference
24 May 2023 – Today, 24 May, the Benelux RFConference takes place, which Noviotech Campus is a partner of. This…
Final call to register for Noviotech Soccer Tournament
It’s going to happen again, Friday afternoon, June 23, 2023, the “Noviotech Soccer Tournament” will take place on Quick’s fields! …
Coming up: Benelux RF Conference
Nijmegen is a leader when it comes to knowledge and innovation in Radio Frequency (RF) Technologies. Bluetooth and WiFi are already…
Looking back on Radboud Festival
Noviotech Campus as a partner of Radboud University, is looking back at Radboud Festival! The celebration of the 100th anniversary…
The Batavierenrace – the largest relay race in the world
The 51st Batavierenrace was held on April 28-29, 2023.The Batavierenrace is the largest relay race in the world. The members…
Koen Dechering tells about artificial intelligence-driven discovery of small molecules
On May 25, during the SMB-meeting From-Molecule-to-Business at Noviotech Campus Koen Dechering, CEO TropIQ Health Sciences will present the parallel track:Artificial…
Settle in the heart of Noviotech Campus
As a first step in the area development we initiated together with MVRDV, we're building The Frontrunner. Check out the...Queen Máxima visits NXP
Her Majesty Queen Máxima will pay a working visit to NXP Semiconductors at Noviotech Campus in Nijmegen on May 25....Opportunities for funding and collaboration
RVO (Rijksdienst voor Ondernemend Nederland) has published two funding programs, which may be of interest to members of the semicon-network:...Medical technology secures future of care
From an artificial pancreas to needle-free injections, medical technology is making healthcare more efficient and allowing people to live healthier...Podcast on NXP’s progress towards the ESG goals
NXP’s Chief Sustainability Officer, Jennifer Wuamett, joins the latest episode of the Smarter World Podcast to discuss NXP’s recently released Corporate...Nexperia announces an environmentally friendly energy harvesting solution
Nexperia, the Noviotech Campus-based expert in essential semiconductors, recently announced an expansion to its range of Power ICs with Energy…
Royal Distinctions for seven Radboudians
Apr 28, 2023 On the occasion of King’s Day this year, seven employees of our partners Radboud University and Radboudumc…
Nijmegen presents Health and High Tech Action Plan
The health and high tech economic clusters are important drivers of the regional economy, with an impact on (inter)national level….
Radboudumc pushes for specialized pediatric trauma care
Radboudumc is launching a Center of Expertise for Child Trauma and Burn Care. The center will provide special care for…
100 Radboud Gestures
Apr 25, 2023 In the week of 8 to 14 May, our partner Radboud University will surprise Nijmegen and its…
Parkinson Weekend Nijmegen
Save the Dates: During the weekend of September 22-24, the city of Nijmegen will be completely dedicated to Parkinson’s disease….
Noviotech Campus signed Human Capital Agreement
13 April 2023 – Provincie Gelderland, The Economic Board and Groene Metropoolregio Arnhem-Nijmegen (GMR) – regio Arnhem Nijmegen signed the…
New Community for Health & High Tech, says Yvette Akkermans
12 April 2023 – Novio Tech Campus in Nijmegen, the capital of Health & High Tech Innovation is increasingly cooperating…
Column by Bert Krikke on AI in the latest ”Ondernemersbelang”
6 April 2023 – Column by Bert Krikke, Director Noviotech Campus on AI (Artificial-Intelligence) in the latest ”Ondernemersbelang”. Of course,…
Chip design key position in Dutch and European Semiconductor
6 April 2023 – The Netherlands has achieved a global top position within the semiconductor equipment industry. ⚡ The second…
Interview Dolf Jansen and Bert Krikke at Health Valley event 2023
5 April 2023 - Recently at the Health Valley event 2023, Bert Krikke talks to Dolf Janssen about the Health...Benelux RF Conference on 24 May 2023
4 April 2023 - Are you a Community member, located at Noviotech Campus and you would like to visit the...Nijmegen region desperately needs international talent
31 March 2023 – The labor market is not in balance, according to alderman John Brom. He talks about the…
Bert Krikke ”Op de bank” bij Dolf Jansen at the Health Valley event
29 March 2023 – The Health Valley Event (HVE) on 30 March 2023 in Pathé Nijmegen is the leading healthcare…
Medip Analytics: A clear overview of what health technology can do for you
28 March 2023 – ”Medip Analytics is really taking off now,” says Tim Govers, CEO, HTA Expert and Business developer….
More Women in Tech needed
23 March 2023 – Karla Swierenga, working as Product Quality Director at NXP Semiconductors and Mitra Gilasgar, working at Ampleon,…
Klokhuis Science Award 2023 for Radboudumc
20 March 2023 – The Klokhuis Science Award was announced during the eighth edition of InScience 2023 by Klokhuis presenter…
When your world turns dark & How to bring light into the dark
22 March 2023 – Joyce de Ruiter, Ambassador of the Usher Syndrome Foundation, was sixteen years old when her parents…
How to address climate change – Shell’s proposal
22 March 2023 – Perhaps everyone is looking at how to combat global warming? Shell proposes to explore carbon capture,…
ChatGPT can stimulate innovation within your organisation
21 March 2023 – ChatGPT took the internet by storm. The chatbot, powered by OpenAI’s GPT-3 large language model, impressed…
Settle in the heart of Noviotech Campus
21 March 2023 Settle in the heart of Noviotech Campus! At Noviotech Campus, a 610 m2 floor is available for rent…
An inspiring day at High Tech Campus Eindhoven & Automotive Campus Helmond
15 March 2023 – A fantastic inspiring day! The Noviotech Campus team together with with colleagues from Oost NL & Gemeente Nijmegen visited…
Another Role it Out training at Noviotech Campus
16 March 2023 – Today Noviotech Campus and Kadans Science Partner were hosting and facilitating the Presentation Skill training from…
Development of NS Station Goffert-Noviotech Campus surroundings
16 March 2023 – Interesting article in De Gelderlander last week about the development of the NS Goffert Station surroundings. Noviotech Campus…
Want to know how technology can improve health care?
07 March 2023 – During the upcoming Health Valley event (30 March), Maroeska Rovers will provide the plenary program and…
Tracking tuberculosis: PinkRF knows preceisly how, when and where
9 March 2023 – PinkRF, based at our Noviotech Campus since 2016, is moving forward rapidly. Klaus Werner, CEO and…
Medip Analytics established at Noviotech Campus
1 March 2023 – We are pleased to announce that today, 1st March 2023, Medip Analytics opened their new office at the Noviotech…
Role it Out role models training at Noviotech Campus
28 February 2023 – Role It Out role models completed their first training about Social Media at the Noviotech Campus….
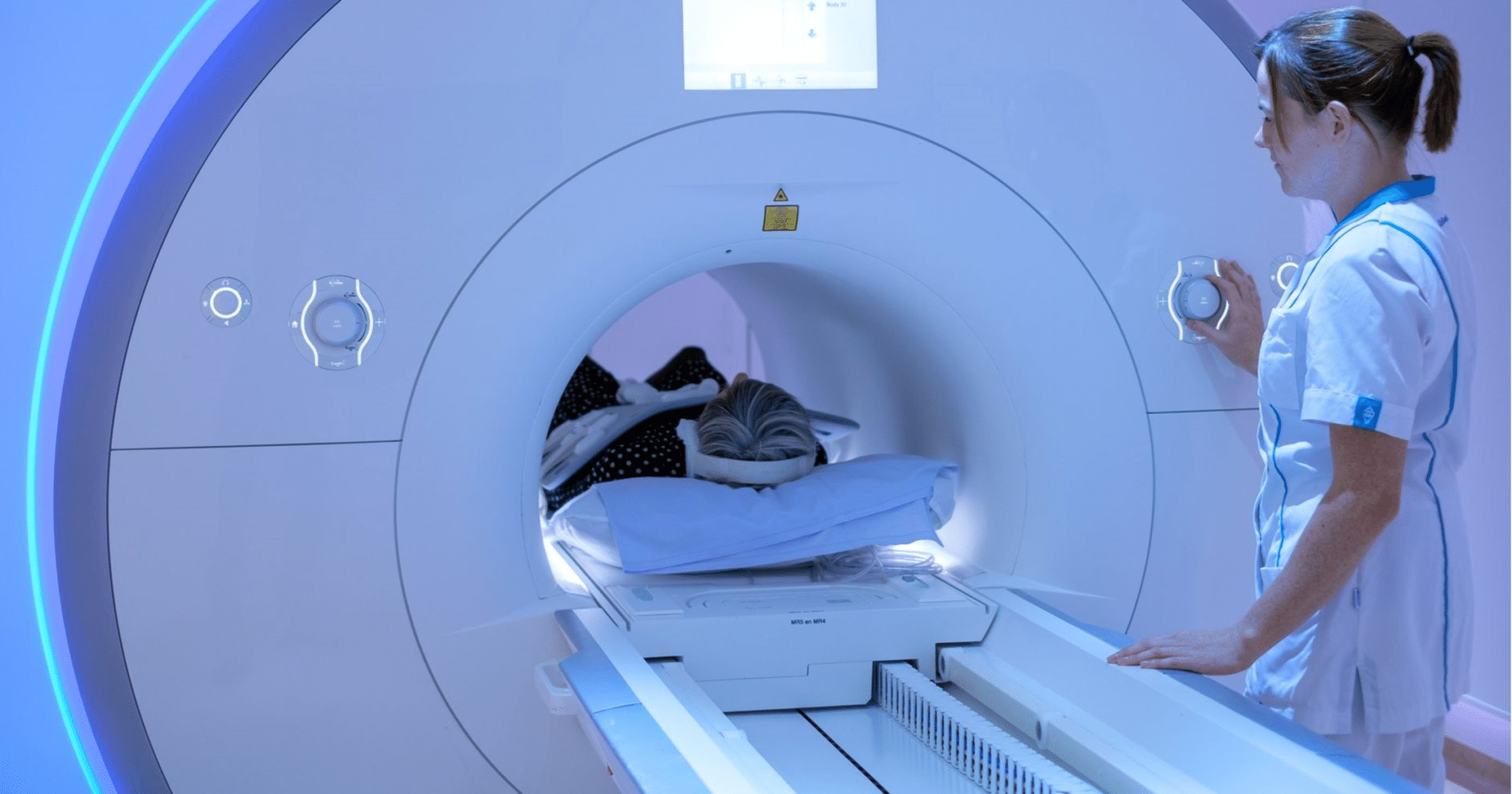
Strongest MRI scanner used for patients in the world will be built in Nijmegen
22 February 2023 – This week the Donders Institute for Brain, Cognition and Behaviour of Radboud University Nijmegen and Radboudumc announced…
Looking back at the Regional Congress Green Metropolitan 2023
22 February 2023 – Noviotech Campus attended the regional conference Green Metropolitan Region Arnhem-Nijmegen (GMR) – Arnhem Nijmegen Region in…
Big Data robot lab to be established at Noviotech Campus
20 February 2023 – The Big Data Robot lab is being developed in cooperation with Fontys University of Applied Sciences…
Chip manufacturer NXP seeks power from its own ‘solar farm’ and wants to buy power from neighbors
16 February 2023 – Not even a split second chip manufacturer NXP survives without power! If there’s no power for…
”De beleving staat centraal” @ Noviotech Campus
15 February 2023 – At Noviotech Campus, on the western side of Nijmegen, the contours of a Health & High Tech…
Inscience Festival
14 February 2023 – InScience, International Science Film Festival Nijmegen is one of the largest international science film festivals in…
Winners of Circular Innovation Top 20
9 February 2023 – This year the Circular Innovation Top 20 was held at Noviotech Campus! The winners of the…
Money Meets Ideas
7 February 2023 Under the name Money Meets Ideas (www.moneymeetsideas.nl), where Rabobank (www.rabobank.nl) and KplusV (www.kplusv.nl) annually organize 20-25 meetings…
Get this book for free: Holding still, together. Parkinson in Beeld.
26 January 2023 This book provides unique insights into how health professionals and people with Parkinson’s disease shape care together….
Sign up for Gelderland Circular Innovation Top 20
23 January 2023 The final of the Gelderland Circular Innovation Top 20 will be on February 9 at 4:00 PM…
The new Brochure Science & Innovation Parks and Campuses in East Netherlands is out!
22 Januray 2023 Of course Campus Nijmegen (Noviotech Campus and Campus Heijendaal) is one of the mentioned parties. Once again…
NXP uses UWB for Smart Homes: Make Smart Homes more intuitive
20 January 2023 NXP looks at UWB as a sensing technology. UWB delivers spatial awareness, which is a new dimension…
Join the smart course in AI at Radboud Academy
20 January 2023 Everyone has to deal with Artificial Intelligence, or AI. In fact: you are probably already dealing with…
AI research Radboud university medical center gets huge boost
10 January 2023 – As many as four completely new AI labs will start within Radboud university medical center with…
Rest, routine and regularity: benefits for every baby
10 January 2023 – Prevention is better than cure, so it is said in the medical world. And according to…
RF Knowledge Lab
9 Januari 2023 – Radio Frequency Technology: to heat, sense, detect and connect. Offering solutions to societal challenges you probably…
Tinybots ”We’re here to make history, not a quick buck”
29 December 2022 – What is the key to success? Is it about personal traits, like talent, intelligence or creativity…
Anna Walesieniuk – meaningful work in exciting times
The Health & High Tech industries are still seen as predominantly male, but in fact women are paving the way…
The Battle for Talent – Bert Krikke
22 December 2022 – The Battle for Talent! In an aging society, there is undeniable competition among companies to retain…
Enzyre uses investments to save thousands on healthcare
22 December 2022 Enzyre uses investments to save thousands on healthcare According to Guido Maertens, co-founder and CEO of Enzyre,…
Lifeport Welcome Center at Huize Heyendael
21 December 2022 Lifeport Welcome Center (LWC) is the central information point in the Arnhem-Nijmegen region where international knowledge workers…
Role It Out Training Program
20 December 2022 The first official Role It Out Training Program is scheduled and participants are registered!! 🚀 It is…
Inspiring story of Kasia Nowak, Technical Director Nexperia
19 December 2022 Paving the way The Health & High Tech industries are still seen as predominantly male, but in…
Holland High Tech pavilion at SEMICON Japan 2022
13 December 2022 In partnership with the Netherlands Embassy in Tokyo, we are organizing the Holland High Tech pavilion at…
Transatlantic Connections Award – application open!
7 december 2022 Are you the next emerging biotech entrepreneur of the Netherlands or Massachusetts? Do you want to have…
New analysis on the Healthcare, IT, Software & Technology market
2 december 2022 Aeternus, supporting partner of Briskr and specialised in company valuation, has published a new analysis on the…
Winkelsteeg: Our city neighbors are expanding!
30 November 2022 Winkelsteeg: Our city neighbors are expanding with several thousand homes and numerous new businesses. Possibly by the…
Noviotech Campus nominated as a sustainable business park!
24 November 2022 Noviotech Campus in the Top 3 of ”Most Climate Adaptive and Nature Inclusive Business Park of the…
Grand Opening building NTS Optel
26 September 2022 On Friday 23th September 2022, Alderman of Economic Affairs John Brom officially opened the new building on…
Showing women why it’s great to work in tech
19 August 2022 How can we help women find out about the possibilities in the tech sector? Lisa Zijm and…
Fighting malaria is fighting poverty
20 May 2022 Diseases that spread across the globe come and go, but malaria has been around for thousands of…
Meniscus prosthesis receives €2.5 million EU grant
10 May 2022 The European Commission is awarding a €2.5 million grant to ATRO Medical for the accelerated development of…
”Spontaneous encounters are not so spontaneous at all”
1 November 2022 Yvette Akkermans: “As a community manager, my job is to connect people. A campus like Noviotech Campus…
Exclusive: Brave and bold in the battle against fake news and conspiracy theories
He got a pie and the word “murderer” thrown in his face recently in Bruges. On social media it was…
Exclusive: Brave and bold in the battle against fake news and conspiracy theories
He got a pie and the word “murderer” thrown in his face recently in Bruges. On social media it was…
Pulse.Magazine #2 – The Future of Health & High Tech
What does the future of health & high tech look like? And what do these industries mean to us: people,…
Bertine Lahuis: “Are we asking the right questions about our future?”
As a mother of two, I see my children searching for meaning. And that’s not surprising because, in our constantly…
Noviotech Campus presented: the Vibe of the Future Festival 2022
With digital biomarkers, you can measure health more objectively. This gives doctors a better picture of a patient. The Nijmegen-based company Orikami develops such biomarkers for practical use, including in cooperation with Radboudumc and Radboud University.
Pulse.Magazine #1 – The unexpected impact of art on health
The first edition Noviotech Campus, with inspiring stories of Health & High Tech. Read full magazine online!Pulse.Magazine #3 – Unexpected intersections in Health & High Tech
Unexpected Intersections in Health & High Tech This third edition of Pulse Magazine explores the power of serendipity in driving…

Pulse, Noviotech Campus’s flagship magazine, shines a light on innovators and entrepreneurs driving positive change. We share stories of visionaries who push boundaries and propel society forward, celebrating entrepreneurship in its purest form.
Through diverse perspectives, Pulse explores the true meaning of innovation and its impact on individuals and communities. Join us as we uncover groundbreaking research and ideas, showcasing the transformative power of human ingenuity.
Podcasts
What’s next in AI, can medical technology improve equality within our society and how does the future of chemistry look like? These podcasts are setting the tone – don’t miss out on them!

AI for Life – Adaptive Learning
How can data and smart technology improve the educational system? A podcast about the application of AI to balance the needs of an individual student with those of the class.

Radboud Reflects – Put Big Tech on a leash?
With fake news, polarising algorythms and inappropriate advertisements, the internet is under pressure. Is it time to restrain the Big Tech industry?

The Huberman Lab – from Andrew Huberman
Andrew Huberman, neuroscientist at Stanford University, discusses the impact of the brain and its connections to our perceptions, behavior and health.
Clips
// health & high tech in the picture
Nijmegen: city of health and high tech